gburdell
Member
Big Time Noob here, with a question about reconstituting NAD+ purchased from QSC about a month ago. I'd post in the QSC UGL thread where there's been some discussion of this, but again, I'm a Big Time Noob. My apologies for the length of this post, if I'd had more time I would have written something shorter.
The QSC NAD+ came as a kit of 140mg vials, physically bigger than what I'm used to for HGH or peptides, same height, but larger diameter. I reconstituted seven vials with 1.4ml of BAC per vial to make 7ml of a 100mg/ml solution of which I injected 1ml IM to the lateral thigh. It hurt like a mofo, worse than test PIP. A rash presented at the injection site within a couple of hours. Pain lasted for almost a week, with walking being especially painful. Obviously this wasn't fun, but it was also out of character from anything I'd experienced with QSC product -- everything else worked well.
Looking at the QSC forum, it's clear that it wasn't just me, most everyone had a similar or worse reaction, with some having fevers. If I weren't so stubborn -- and maybe stupid, I would have trashed it all right there, but I didn't.
I filtered the remaining 6ml of solution through a 22μm filter, and then tried injecting that. It seems that that stopped the rash, and reduced the PIP a bit, but it was still there. Forgive the pun, but this was not a good solution.
Reading along in the QSC thread, @gettinginshape tested his similar solution for pH with strips and was seeing something between 3-4. This got me thinking that the PIP may be related to the acidity.
I tested my solution and saw 2.5 on a cheapo Amazon pH meter; per Google, NAD+ doesn't survive long in a very acidic solution, so I pitched what I had. Using my new-found Broscience degree, and despite my failing of chemistry in college, I expect that as pH is logarithmic, that diluting with water won't result in a volume of solution that will be able to be reasonably injected, at least short of taking on a liter of saline with each dose.
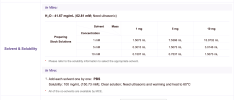
Experimentally, I came up with reconstituting a 140mg vial with 1ml of BAC and 400μl of 8.4% NaOH buffer solution resulting in the 100mg/ml solution I was looking for, but at a much more reasonable pH of 6.5. I injected 1ml of this new solution a few hours ago without any discomfort.
My questions are regarding my Broscience here -- was my thinking generally sound? Anything I should be thinking/doing differently? It seems to work, and unless NaOH damages the NAD+, I believe I've found a reasonable solution -- any thoughts from the more experienced among you?
Separate but related, is acidity an issue with peptides and related injectables? I don't have a ton of experience, but have injected a lot of peptides and similar and never had any reaction anything like this. Do other NAD+ suppliers maybe pre-buffer their NAD+ before lypholyzing it?
The QSC NAD+ came as a kit of 140mg vials, physically bigger than what I'm used to for HGH or peptides, same height, but larger diameter. I reconstituted seven vials with 1.4ml of BAC per vial to make 7ml of a 100mg/ml solution of which I injected 1ml IM to the lateral thigh. It hurt like a mofo, worse than test PIP. A rash presented at the injection site within a couple of hours. Pain lasted for almost a week, with walking being especially painful. Obviously this wasn't fun, but it was also out of character from anything I'd experienced with QSC product -- everything else worked well.
Looking at the QSC forum, it's clear that it wasn't just me, most everyone had a similar or worse reaction, with some having fevers. If I weren't so stubborn -- and maybe stupid, I would have trashed it all right there, but I didn't.
I filtered the remaining 6ml of solution through a 22μm filter, and then tried injecting that. It seems that that stopped the rash, and reduced the PIP a bit, but it was still there. Forgive the pun, but this was not a good solution.
Reading along in the QSC thread, @gettinginshape tested his similar solution for pH with strips and was seeing something between 3-4. This got me thinking that the PIP may be related to the acidity.
I tested my solution and saw 2.5 on a cheapo Amazon pH meter; per Google, NAD+ doesn't survive long in a very acidic solution, so I pitched what I had. Using my new-found Broscience degree, and despite my failing of chemistry in college, I expect that as pH is logarithmic, that diluting with water won't result in a volume of solution that will be able to be reasonably injected, at least short of taking on a liter of saline with each dose.
Experimentally, I came up with reconstituting a 140mg vial with 1ml of BAC and 400μl of 8.4% NaOH buffer solution resulting in the 100mg/ml solution I was looking for, but at a much more reasonable pH of 6.5. I injected 1ml of this new solution a few hours ago without any discomfort.
My questions are regarding my Broscience here -- was my thinking generally sound? Anything I should be thinking/doing differently? It seems to work, and unless NaOH damages the NAD+, I believe I've found a reasonable solution -- any thoughts from the more experienced among you?
Separate but related, is acidity an issue with peptides and related injectables? I don't have a ton of experience, but have injected a lot of peptides and similar and never had any reaction anything like this. Do other NAD+ suppliers maybe pre-buffer their NAD+ before lypholyzing it?